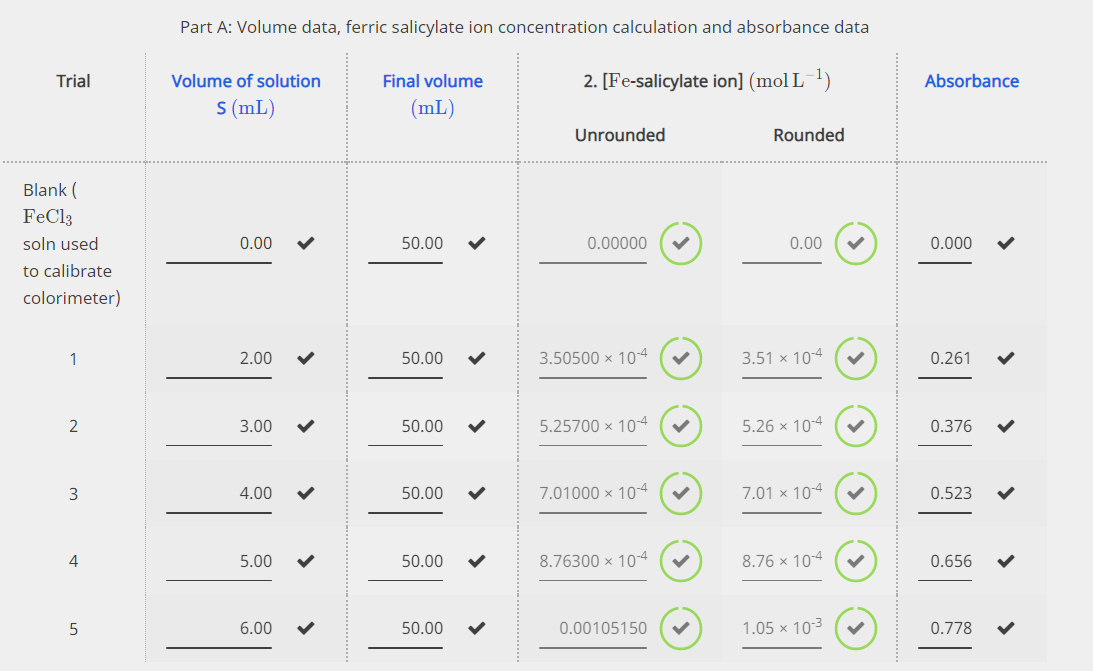
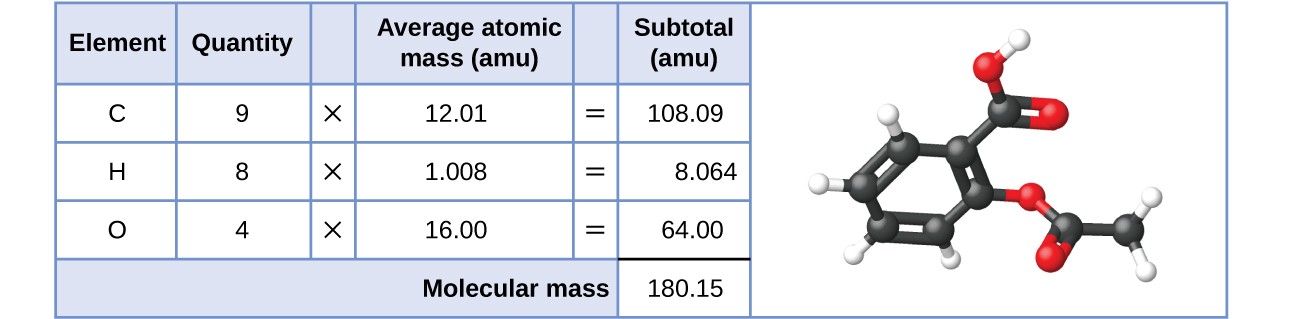
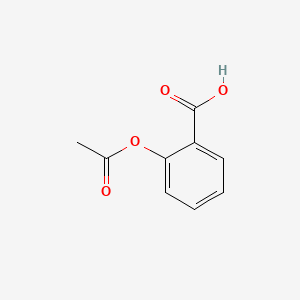
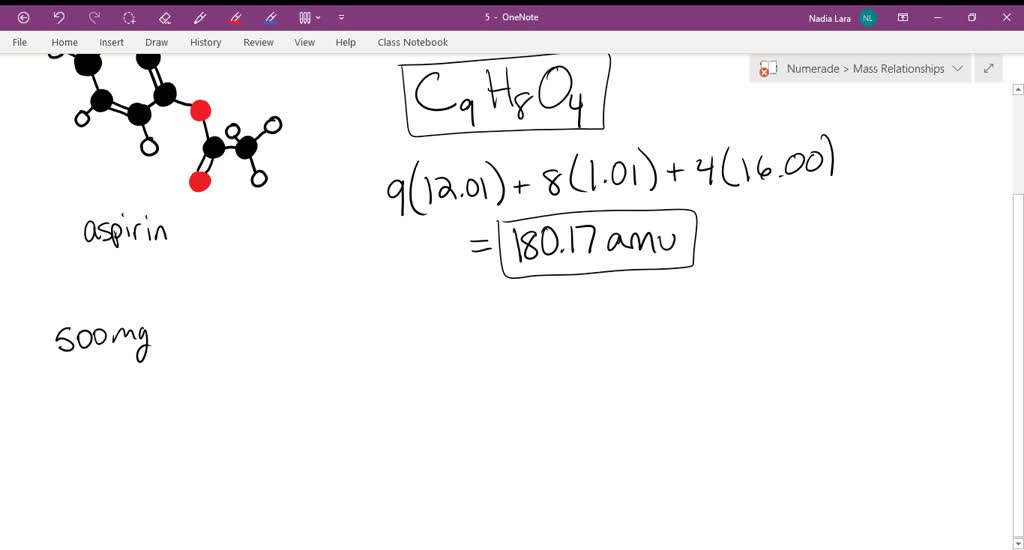
SOLVED:The molecular formula of acetylsalicylic acid (aspirin), one of the most commonly used pain relievers, is C9 H8 O4 a. Calculate the molar mass of aspirin. b. A typical aspirin tablet contains
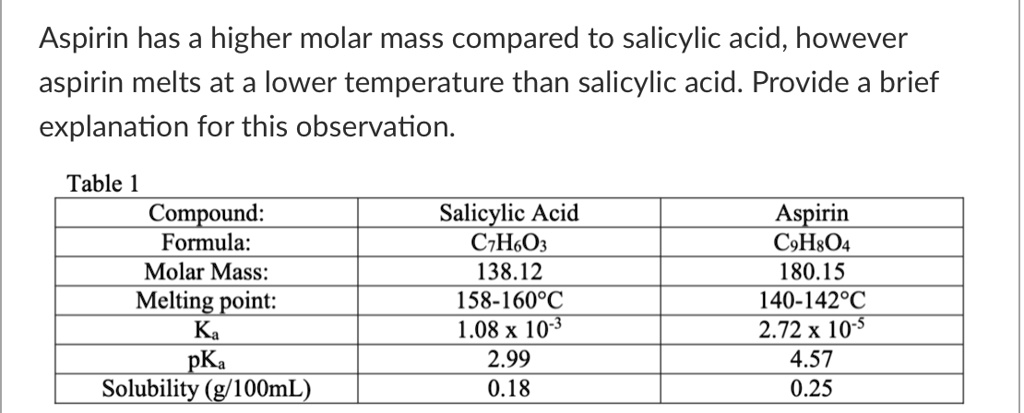
SOLVED: Aspirin has a higher molar mass compared to salicylic acid, however aspirin melts at a lower temperature than salicylic acid. Provide a brief explanation for this observation: Table 1 Compound: Formula:
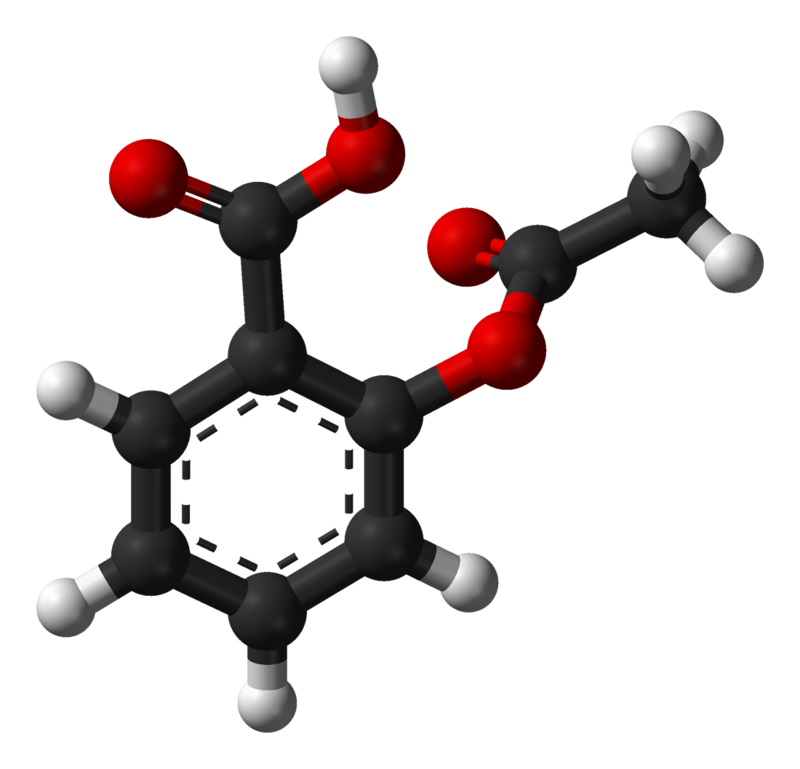
SOLVED:Aspirin can be represented by the adjacent ball-and-stick molecular model. Give the formula for aspirin, and calculate its molecular mass (red =O, gray =C, ivory =H ). How many moles of aspirin

Sketch the molecular structure of acetylsalicylic acid and calculate its molar mass. | Homework.Study.com

Acetyl salicylic acid (mol. wt. = 180) called aspirin is a pain killer with pKa = 6 . It two tablets each of 0.09 gm mass containing aspirin are dissolved in 100 mL solution. Its pH will be:

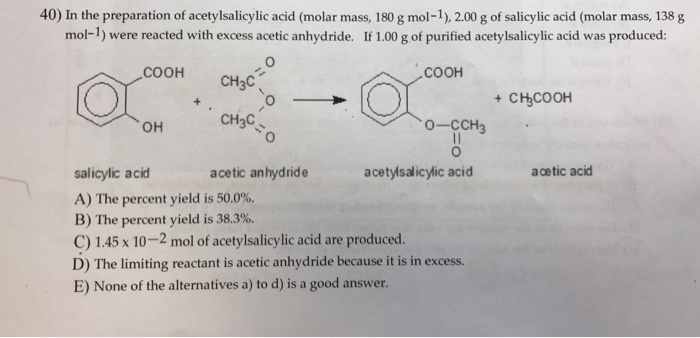
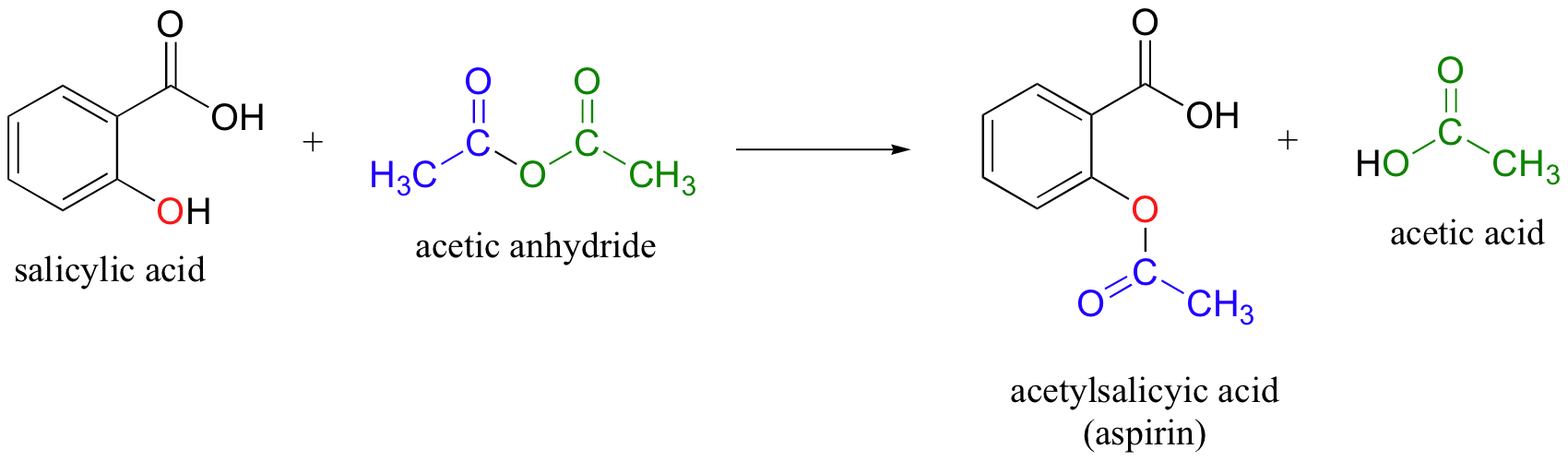
SOLVED: (T) Aspirin (CoHsO+: molar mass = 180.157 glmol) is produced from the reaction of salicylic acid (C-HsOa, molar mass = 138.1207 g/mol) and acetic anhydride (CAHsO3 molar mass 102.0887 g/mol): CzHsO3 +